General workflow
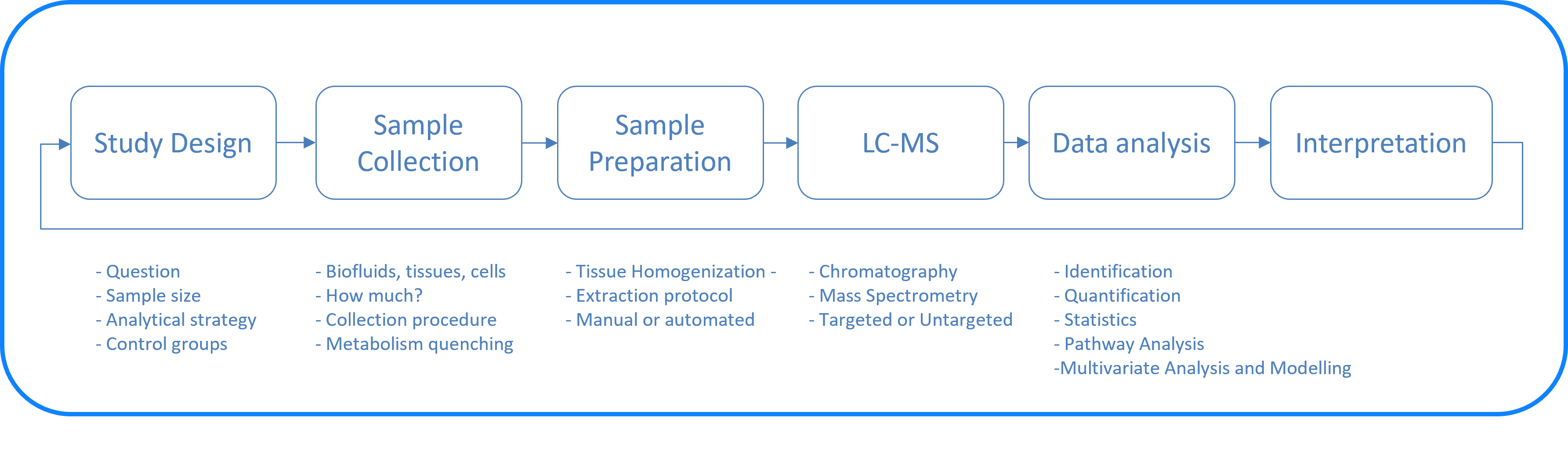
- Study Design: It is important to consider a design that allows to answer the research question based on our current best knowledge. Check the literature, formulate a clear research question, try to mentally simulate the best (and worst) possible outcomes.
- Sample Collection: As metabolism is very fast and continuous process, extra care should be taken to stop metabolism (quenching) once the system is out of its natural state. Different protocols for quenching might be necessary for different applications.
- Sample Preparation: As described below, this is usually done in our labs at FGCZ and in some cases can be done by the users.
- LC/MS measurement: several analytical workflows exist to cover different chemical classes as described briefly below
- Data Analysis: This entails the analysis of raw mass spectrometry files into data tables as a first step. That is usually followed by basic statistical analysis
- Interpretation: This step answers the research question through the acquired metabolomics or lipidomics datasets and the following statistical analysis.
- New questions with new study design: As the process is iterative, usually successful experiments will lead to more targeted questions and hypothesis that can be refined by following the same workflow or performing targeted LC-MS.
- Untargeted metabolomics aims at the detection and relative quantification of polar and charged metabolites and related chemical classes. This covers metabolites involved in glycolysis, TCA, amino acid metabolism, nucleotide metabolism, and related pathways.
- Untargeted Lipidomics aims at the detection and relative quantification of membrane (structural) lipids. This covers phospholipids, sphingolipids, glycerolipids and cholesterol esters. The methods should also cover free fatty acids and acylcarnitines.
- The targeted workflow is more suited where absolute quantificaiton ( e.g pmoles/ml) is required of pre-defined subset of metabolites or lipids. Our current targeted LC-MS portfolio covers short-chain fatty acids, bile acids, modified nucleotides, some bioactive lipids such as retinoic acid and some drugs. For selected projects, method development can be performed. FDA guidelines for bioanalytical method validation are followed to ensure the highest standards of analytical robustness and confidence in both identification and quantification.
